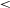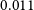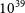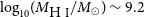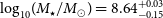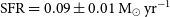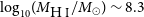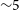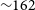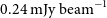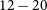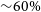14 results
Integrated efficacy analysis from phase 3 studies of investigational microbiome therapeutic, SER-109, in recurrent Clostridioides difficile infection
- Matthew Sims, Michael Silverman, Thomas Louie, Elaine Wang, Colleen Kraft, Mayur Ramesh, Tatiana Bogdanovich, Kelly Brady, David Lombardi, Asli Memisoglu, Ananya De, Brooke Hasson, Christine Lee, Paul Feuerstadt, Darrell Pardi, Colleen Kelly, Peter Daley, Godson Oguchi, Barbara McGovern, Lisa Von Moltke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s5
-
- Article
-
- You have access Access
- Open access
- Export citation
-
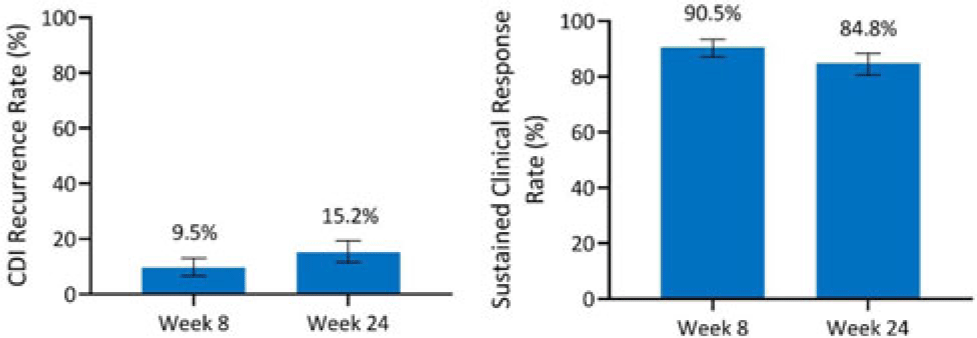
Background: Antibiotics alone are often insufficient to treat recurrent C. difficile infection (rCDI) because they have no activity against C. difficile spores that germinate within a disrupted microbiome. SER-109, an investigational, oral, microbiome therapeutic comprised of purified Firmicutes spores, was designed to reduce rCDI through microbiome repair. We report an integrated efficacy analysis through week 24 for SER-109 from phase 3 studies, ECOSPOR III and ECOSPOR IV. Methods: ECOSPOR III was a randomized, placebo-controlled phase 3 trial conducted at 56 US or Canadian sites that included 182 participants with ≥2 CDI recurrences, confirmed via toxin EIA testing. Participants were stratified by age (<65 years or ≥65 years) and antibiotic regimen (vancomycin, fidaxomicin) and were randomized 1:1 to placebo or SER-109 groups. ECOSPOR IV was an open-label, single-arm study conducted at 72 US or Canadian sites including 263 participants with rCDI enrolled in 2 cohorts: (1) rollover participants from ECOSPOR III who experienced on-study recurrence diagnosed by toxin EIA (n = 29) and (2) participants with ≥1 CDI recurrence (diagnosed by PCR or toxin EIA), inclusive of the current episode (n = 234). In both studies, the investigational product was administered orally as 4 capsules over 3 consecutive days following symptom resolution after standard-of-care antibiotics. The primary efficacy end point was rCDI (recurrent toxin-positive diarrhea requiring treatment) through week 8. Other end points included CDI recurrence rates and safety through 24 weeks. Results: These 349 participants received at least 1 dose of SER-109 in ECOSPOR III or ECOSPOR IV (mean age 64.2; 68.8% female). Overall, 77 participants (22.1%) enrolled with their first CDI recurrence. Four participants received blinded SER-109 in ECOSPOR III followed by a second dose of open-label SER-109 in ECOSPOR IV. Overall, the proportion of participants who received any dose of SER-109 with rCDI at week 8 was 9.5% (33 of 349; 95% CI, 6.6 %–13.0%), and the CDI recurrence rate remained low through 24 weeks (15.2%, 53 of 349; 95% CI, 11.6%–19.4%), corresponding to sustained clinical response rates of 90.5% (95% CI, 87.0%–93.4%) and 84.8% (95% CI, 80.6%–88.4%), respectively (Fig. 1). Most rollover participants (25 of 29, 86.2%) were from the placebo arm; 13.8% had rCDI by week 8. Conclusions: In this integrated analysis, the rates of rCDI were low and durable in participants who received the investigational microbiome therapeutic SER-109, with sustained clinical response rates of 90.5% and 84.8% at weeks 8 and 24, respectively. These data further support the potential benefit of microbiome repair with SER-109 following antibiotics for rCDI to prevent recurrence in high-risk patients.
Financial support: This study was funded by Seres Therapeutics.
Disclosure: None
Integrated safety analysis of phase 3 studies for investigational microbiome therapeutic, SER-109, in recurrent CDI
- Matthew Sims, Charles Berenson, Stuart Cohen, Elaine Wang, Elizabeth Hohmann, Richard Nathan, Alberto Odio, Paul Cook, Kelly Brady, David Lombardi, Asli Memisoglu, Ananya De, Brooke Hasson, Bret Lashner, Louis Korman, Doria Grimard, Juan Carlos Moises Gutierrez, Barbara McGovern, Lisa Von Moltke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s44-s45
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Clostridioides difficile infection (CDI) often recurs in patients aged ≥65 years and those with comorbidities. Clinical trials often exclude patients with history of immunosuppression, malignancy, renal insufficiency, or other comorbidities. In a phase 3 trial (ECOSPOR III), SER-109 was superior to placebo in reducing recurrent CDI (rCDI) risk at week 8 and was well tolerated. We report integrated safety data for SER-109 in a broad patient population through week 24 from phase 3 studies: ECOSPOR III and ECOSPOR IV. Methods: ECOSPOR III was a double-blind, placebo-controlled trial conducted in participants with ≥2 CDI recurrences randomized 1:1 to placebo or SER-109. ECOSPOR IV was an open-label, single-arm study conducted in 263 patients with rCDI enrolled in 2 cohorts: (1) rollover participants from ECOSPOR III with on-study recurrence and (2) participants with ≥1 CDI recurrence, inclusive of the current episode. In both studies, the investigational product was administered as 4 oral capsules over 3 days. Treatment-emergent adverse events (TEAEs) were collected through week 8; serious TEAEs and TEAEs of special interest (ie, bacteremia, abscess, meningitis) were collected through week 24. Results: In total, 349 participants received SER-109 in ECOSPOR III and/or ECOSPOR IV (mean age 64.2; 68.8% female). Chronic diseases included cardiac disease (31.2%), immunocompromised or immunosuppressed (21.2%), diabetes (18.9% ), and renal impairment or failure (13.2%). Overall, 221 (63.3%) of 349 participants who received SER-109 experienced TEAEs through week 24. Most were mild to moderate and gastrointestinal. The most common (>5% of participants) treatment related TEAEs were flatulence, abdominal pain and distension, decreased appetite, constipation, nausea, fatigue, and diarrhea. No participants experienced a treatment-related TEAE leading to study withdrawal. Invasive infections were observed in 28 participants (8%); those with identified pathogens were unrelated to SER-109 species, and all were deemed unrelated to treatment by the investigators. There were 11 deaths (3.2%) and 48 participants (13.8%) with serious TEAEs, none of which were deemed treatment related. There were no clinically important differences in the safety profile across subgroups of sex, race, prior antibiotic regimen, or number of CDI recurrences. No safety signals were observed in participants with renal impairment or failure, diabetes, cardiac disease, or immunocompromised or immunosuppressed individuals. Conclusions: In this integrated analysis of phase 3 trials, SER-109, an investigational microbiome therapeutic, was well tolerated in this vulnerable patient population with prevalent comorbidities. No infections, nor those with identified pathogens, were attributed to SER-109 or product species. This safety profile might be expected because this purified product is composed of spore-forming Firmicutes normally abundant in the healthy microbiome.
Financial support: This study was funded by Seres Therapeutics.
Disclosures: None
The host galaxy of FRB 20171020A revisited
- Karen Lee-Waddell, Clancy W. James, Stuart D. Ryder, Elizabeth K. Mahony, Arash Bahramian, Bärbel S. Koribalski, Pravir Kumar, Lachlan Marnoch, Freya O. North-Hickey, Elaine M. Sadler, Ryan Shannon, Nicolas Tejos, Jessica E. Thorne, Jing Wang, Randall Wayth
-
- Journal:
- Publications of the Astronomical Society of Australia / Volume 40 / 2023
- Published online by Cambridge University Press:
- 11 July 2023, e029
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
The putative host galaxy of FRB 20171020A was first identified as ESO 601-G036 in 2018, but as no repeat bursts have been detected, direct confirmation of the host remains elusive. In light of recent developments in the field, we re-examine this host and determine a new association confidence level of 98%. At 37 Mpc, this makes ESO 601-G036 the third closest FRB host galaxy to be identified to date and the closest to host an apparently non-repeating FRB (with an estimated repetition rate limit of
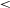 $<$
$<$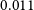 $0.011$ bursts per day above
$0.011$ bursts per day above 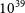 $10^{39}$ erg). Due to its close distance, we are able to perform detailed multi-wavelength analysis on the ESO 601-G036 system. Follow-up observations confirm ESO 601-G036 to be a typical star-forming galaxy with H i and stellar masses of
$10^{39}$ erg). Due to its close distance, we are able to perform detailed multi-wavelength analysis on the ESO 601-G036 system. Follow-up observations confirm ESO 601-G036 to be a typical star-forming galaxy with H i and stellar masses of 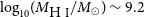 $\log_{10}\!(M_{\rm{H\,{\small I}}} / M_\odot) \sim 9.2$ and
$\log_{10}\!(M_{\rm{H\,{\small I}}} / M_\odot) \sim 9.2$ and 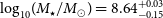 $\log_{10}\!(M_\star / M_\odot) = 8.64^{+0.03}_{-0.15}$, and a star formation rate of
$\log_{10}\!(M_\star / M_\odot) = 8.64^{+0.03}_{-0.15}$, and a star formation rate of 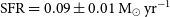 $\text{SFR} = 0.09 \pm 0.01\,{\rm M}_\odot\,\text{yr}^{-1}$. We detect, for the first time, a diffuse gaseous tail (
$\text{SFR} = 0.09 \pm 0.01\,{\rm M}_\odot\,\text{yr}^{-1}$. We detect, for the first time, a diffuse gaseous tail (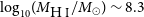 $\log_{10}\!(M_{\rm{H\,{\small I}}} / M_\odot) \sim 8.3$) extending to the south-west that suggests recent interactions, likely with the confirmed nearby companion ESO 601-G037. ESO 601-G037 is a stellar shred located to the south of ESO 601-G036 that has an arc-like morphology, is about an order of magnitude less massive, and has a lower gas metallicity that is indicative of a younger stellar population. The properties of the ESO 601-G036 system indicate an ongoing minor merger event, which is affecting the overall gaseous component of the system and the stars within ESO 601-G037. Such activity is consistent with current FRB progenitor models involving magnetars and the signs of recent interactions in other nearby FRB host galaxies.
$\log_{10}\!(M_{\rm{H\,{\small I}}} / M_\odot) \sim 8.3$) extending to the south-west that suggests recent interactions, likely with the confirmed nearby companion ESO 601-G037. ESO 601-G037 is a stellar shred located to the south of ESO 601-G036 that has an arc-like morphology, is about an order of magnitude less massive, and has a lower gas metallicity that is indicative of a younger stellar population. The properties of the ESO 601-G036 system indicate an ongoing minor merger event, which is affecting the overall gaseous component of the system and the stars within ESO 601-G037. Such activity is consistent with current FRB progenitor models involving magnetars and the signs of recent interactions in other nearby FRB host galaxies.
Healthcare resource utilization in a phase 3 trial of SER-109 in patients with recurrent Clostridioides difficile infection
- Stuart Cohen, Thomas Louie, Charles Berenson, Alpesh Amin, David Lombardi, Sissi Pham, Shirley Huang, Elaine Wang, Brooke Hasson, Barbara McGovern, Lisa Von Moltke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s73
-
- Article
-
- You have access Access
- Open access
- Export citation
-
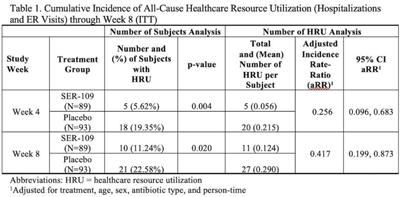
Background: The estimated economic cost of Clostridioides difficile infection (CDI) is $5.4 billion annually, primarily attributed to acute-care costs. We previously reported data from ECOSPOR III that SER-109, an investigational oral microbiome therapeutic, was superior to placebo in reducing recurrent CDI (rCDI) in adults at 8 weeks after treatment, with a 68% relative risk reduction. Adults with rCDI have more hospitalizations and emergency room (ER) visits (defined herein as healthcare resource utilization, HRU) compared to those without recurrence. Thus, we evaluated incidence of HRU. Methods: Adults with rCDI (≥3 episodes in 12 months) were screened at 56 US and Canadian sites and were randomized 1:1 to SER-109 (4 capsules × 3 days) or placebo following resolution of CDI with standard-of-care CDI antibiotics. The primary end point was rCDI at 8 weeks. Exploratory end points included cumulative incidence of hospitalizations through 24 weeks after treatment. Here, we report cumulative incidence of all-cause HRU through 8 weeks after treatment. Results: In total, 281 patients were screened and 182 were randomized (59.9% female; mean age 65.5 years; 98.9% outpatient). Overall, 31 patients (17%) had 38 hospitalizations or ER visits through week 8 (11 events in 10 SER-109 patients and 27 events in 21 placebo patients) (Table 1). The cumulative incidence of HRU was lower in SER-109–treated patients compared to placebo at both weeks 4 and 8 with most events (65.8%) recorded within 4 weeks after treatment. The adjusted HRU incidence rate (by person time, age, sex, and antibiotic use) was also lower in SER-109–treated patients compared to placebo at weeks 4 and 8 (0.256 [95% CI, 0.096–0.683] versus 0.417 [95% CI, 0.199–0.873], respectively). Conclusions: SER-109–treated patients had less HRU compared to placebo patients through 8 weeks after treatment in this mostly outpatient population. These data suggest a potential benefit of SER-109 in reducing HRU, thus lowering the healthcare burden of rCDI.
Funding: Seres Therapeutics
Disclosures: None
The ASKAP Variables and Slow Transients (VAST) Pilot Survey
- Part of
- Tara Murphy, David L. Kaplan, Adam J. Stewart, Andrew O’Brien, Emil Lenc, Sergio Pintaldi, Joshua Pritchard, Dougal Dobie, Archibald Fox, James K. Leung, Tao An, Martin E. Bell, Jess W. Broderick, Shami Chatterjee, Shi Dai, Daniele d’Antonio, Gerry Doyle, B. M. Gaensler, George Heald, Assaf Horesh, Megan L. Jones, David McConnell, Vanessa A. Moss, Wasim Raja, Gavin Ramsay, Stuart Ryder, Elaine M. Sadler, Gregory R. Sivakoff, Yuanming Wang, Ziteng Wang, Michael S. Wheatland, Matthew Whiting, James R. Allison, C. S. Anderson, Lewis Ball, K. Bannister, D. C.-J. Bock, R. Bolton, J. D. Bunton, R. Chekkala, A. P Chippendale, F. R. Cooray, N. Gupta, D. B. Hayman, K. Jeganathan, B. Koribalski, K. Lee-Waddell, Elizabeth K. Mahony, J. Marvil, N. M. McClure-Griffiths, P. Mirtschin, A. Ng, S. Pearce, C. Phillips, M. A. Voronkov
-
- Journal:
- Publications of the Astronomical Society of Australia / Volume 38 / 2021
- Published online by Cambridge University Press:
- 12 October 2021, e054
-
- Article
-
- You have access Access
- HTML
- Export citation
-
The Variables and Slow Transients Survey (VAST) on the Australian Square Kilometre Array Pathfinder (ASKAP) is designed to detect highly variable and transient radio sources on timescales from 5 s to
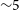 $\sim\!5$
yr. In this paper, we present the survey description, observation strategy and initial results from the VAST Phase I Pilot Survey. This pilot survey consists of
$\sim\!5$
yr. In this paper, we present the survey description, observation strategy and initial results from the VAST Phase I Pilot Survey. This pilot survey consists of 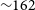 $\sim\!162$
h of observations conducted at a central frequency of 888 MHz between 2019 August and 2020 August, with a typical rms sensitivity of
$\sim\!162$
h of observations conducted at a central frequency of 888 MHz between 2019 August and 2020 August, with a typical rms sensitivity of 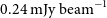 $0.24\ \mathrm{mJy\ beam}^{-1}$
and angular resolution of
$0.24\ \mathrm{mJy\ beam}^{-1}$
and angular resolution of 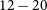 $12-20$
arcseconds. There are 113 fields, each of which was observed for 12 min integration time, with between 5 and 13 repeats, with cadences between 1 day and 8 months. The total area of the pilot survey footprint is 5 131 square degrees, covering six distinct regions of the sky. An initial search of two of these regions, totalling 1 646 square degrees, revealed 28 highly variable and/or transient sources. Seven of these are known pulsars, including the millisecond pulsar J2039–5617. Another seven are stars, four of which have no previously reported radio detection (SCR J0533–4257, LEHPM 2-783, UCAC3 89–412162 and 2MASS J22414436–6119311). Of the remaining 14 sources, two are active galactic nuclei, six are associated with galaxies and the other six have no multi-wavelength counterparts and are yet to be identified.
$12-20$
arcseconds. There are 113 fields, each of which was observed for 12 min integration time, with between 5 and 13 repeats, with cadences between 1 day and 8 months. The total area of the pilot survey footprint is 5 131 square degrees, covering six distinct regions of the sky. An initial search of two of these regions, totalling 1 646 square degrees, revealed 28 highly variable and/or transient sources. Seven of these are known pulsars, including the millisecond pulsar J2039–5617. Another seven are stars, four of which have no previously reported radio detection (SCR J0533–4257, LEHPM 2-783, UCAC3 89–412162 and 2MASS J22414436–6119311). Of the remaining 14 sources, two are active galactic nuclei, six are associated with galaxies and the other six have no multi-wavelength counterparts and are yet to be identified.
Efficacy and Safety of Investigational Microbiome Drug SER-109 for Treatment of Recurrent Clostridioides difficile Infection
- Barbara McGovern, Mathew Sims, Colleen Kraft, Elaine Wang, Kelly Brady, Christopher Ford, O’Brien Edward, Mary-Jane Lombardo, Jennifer Wortman, Kevin Litcofsky, Jennifer Mahoney, Christopher McChalicher, Jonathan Winkler, Sarah Garant, John Aunins, Matthew Henn, Lisa von Moltke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s5
-
- Article
-
- You have access Access
- Open access
- Export citation
-
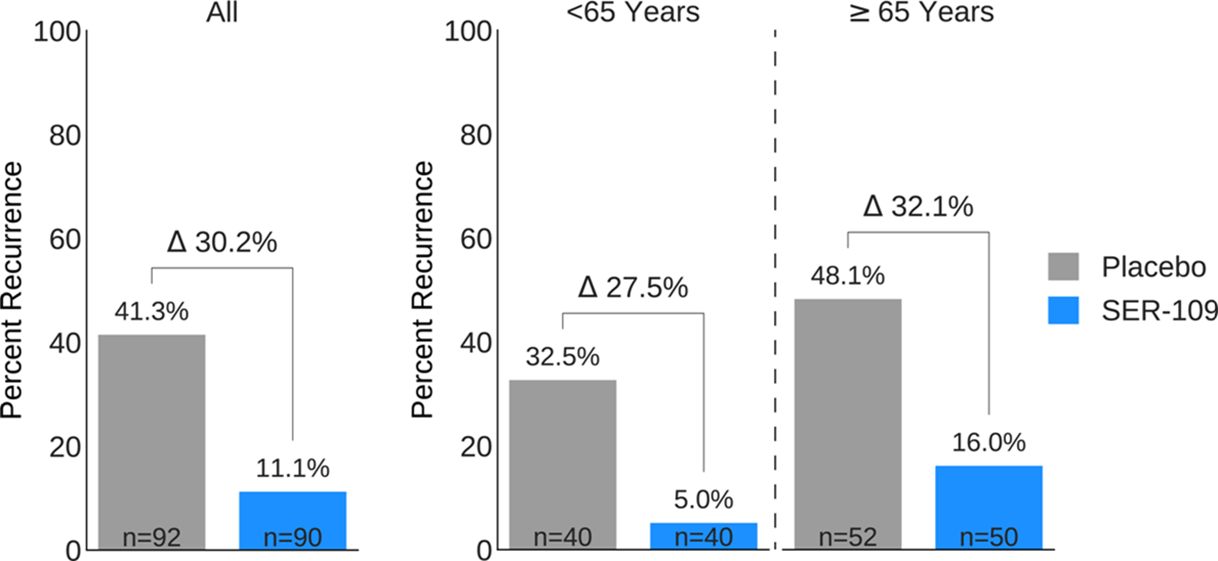
Background: Antibiotics targeted against Clostridioides difficile bacteria are necessary, but insufficient, to achieve a durable clinical response because they have no effect on C. difficile spores that germinate within a disrupted microbiome. ECOSPOR-III evaluated SER-109, an investigational, biologically derived microbiome therapeutic of purified Firmicute spores for treatment of rCDI. Herein, we present the interim analysis in the ITT population at 8 and 12 weeks. Methods: Adults ≥18 years with rCDI (≥3 episodes in 12 months) were screened at 75 US and CAN sites. CDI was defined as ≥3 unformed stools per day for <48 hours with a positive C. difficile assay. After completion of 10–21 days of vancomycin or fidaxomicin, adults with symptom resolution were randomized 1:1 to SER-109 (4 capsules × 3 days) or matching placebo and stratified by age (≥ or <65 years) and antibiotic received. Primary objectives were safety and efficacy at 8 weeks. Primary efficacy endpoint was rCDI (recurrent toxin+ diarrhea requiring treatment); secondary endpoints included efficacy at 12 weeks after dosing. Results: Overall, 287 participants were screened and 182 were randomized (59.9% female; mean age, 65.5 years). The most common reason for screen failure was a negative C. difficile toxin assay. A significantly lower proportion of SER-109 participants had rCDI after dosing compared to placebo at week 8 (11.1% vs 41.3%, respectively; relative risk [RR], 0.27; 95% confidence interval [CI], 0.15–0.51; p-value <0.001). Efficacy rates were significantly higher with SER-109 vs placebo in both stratified age groups (Figure 1). SER-109 was well-tolerated with a safety profile similar to placebo. The most common treatment-emergent adverse events (TEAEs) were gastrointestinal and were mainly mild to moderate. No serious TEAEs, infections, deaths, or drug discontinuations were deemed related to study drug. Conclusions: SER-109, an oral live microbiome therapeutic, achieved high rates of sustained clinical response with a favorable safety profile. By enriching for Firmicute spores, SER-109 achieves high efficacy while mitigating risk of transmitting infectious agents, beyond donor screening alone. SER-109 represents a major paradigm shift in the clinical management of patients with recurrent CDI. Clinicaltrials.gov Identifier NCT03183128. These data were previously presented as a late breaker at American College of Gastroenterology 2020.
Funding: Seres Therapeutics
Disclosures: None
Figure 1.
A Machine-Learning Approach For Predicting Antibiotic Resistance in Pseudomonas aeruginosa
- Reed Magleby, Matthew Simon, David Calfee, Philip Zachariah, Bevin Cohen, Casey Cazer, Fei Wang, Elaine Larson
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s96-s97
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background:Pseudomonas aeruginosa is an important nosocomial pathogen associated with intrinsic and acquired resistance mechanisms to major classes of antibiotics. To better understand clinical risk factors for drug-resistant P. aeruginosa infection, decision-tree models for the prediction of fluoroquinolone and carbapenem-resistant P. aeruginosa were constructed and compared to multivariable logistic regression models using performance characteristics. Methods: In total, 5,636 patients admitted to 4 hospitals within a New York City healthcare system from 2010 to 2016 with blood, respiratory, wound, or urine cultures growing PA were included in the analysis. Presence or absence of drug-resistance was defined using the first culture of any source positive for P. aeruginosa during each hospitalization. To train and validate the prediction models, cases were randomly split (60 of 40) into training and validation datasets. Clinical decision-tree models for both fluoroquinolone and carbapenem resistance were built from the training dataset using 21 clinical variables of interest, and multivariable logistic regression models were built using the 16 clinical variables associated with resistance in bivariate analyses. Decision-tree models were optimized using K-fold cross validation, and performance characteristics between the 4 models were compared. Results: From 2010 through 2016, prevalence of fluoroquinolone and carbapenem resistance was 32% and 18%, respectively. For fluoroquinolone resistance, the logistic regression algorithm attained a positive predictive value (PPV) of 0.57 and a negative predictive value (NPV) of 0.73 (sensitivity, 0.27; specificity, 0.90) and the decision-tree algorithm attained a PPV of 0.65 and an NPV of 0.72 (sensitivity 0.21, specificity 0.95). For carbapenem resistance, the logistic regression algorithm attained a PPV of 0.53 and a NPV of 0.85 (sensitivity 0.20, specificity 0.96) and the decision-tree algorithm attained a PPV of 0.59 and an NPV of 0.84 (sensitivity 0.22, specificity 0.96). The decision-tree partitioning algorithm identified prior fluoroquinolone resistance, SNF stay, sex, and length-of-stay as variables of greatest importance for fluoroquinolone resistance compared to prior carbapenem resistance, age, and length-of-stay for carbapenem resistance. The highest-performing decision tree for fluoroquinolone resistance is illustrated in Fig. 1. Conclusions: Supervised machine-learning techniques may facilitate prediction of P. aeruginosa resistance and risk factors driving resistance patterns in hospitalized patients. Such techniques may be applied to readily available clinical information from hospital electronic health records to aid with clinical decision making.
Funding: None
Disclosures: None
An ultra-wide bandwidth (704 to 4 032 MHz) receiver for the Parkes radio telescope
- George Hobbs, Richard N. Manchester, Alex Dunning, Andrew Jameson, Paul Roberts, Daniel George, J. A. Green, John Tuthill, Lawrence Toomey, Jane F. Kaczmarek, Stacy Mader, Malte Marquarding, Azeem Ahmed, Shaun W. Amy, Matthew Bailes, Ron Beresford, N. D. R. Bhat, Douglas C.-J. Bock, Michael Bourne, Mark Bowen, Michael Brothers, Andrew D. Cameron, Ettore Carretti, Nick Carter, Santy Castillo, Raji Chekkala, Wan Cheng, Yoon Chung, Daniel A. Craig, Shi Dai, Joanne Dawson, James Dempsey, Paul Doherty, Bin Dong, Philip Edwards, Tuohutinuer Ergesh, Xuyang Gao, JinLin Han, Douglas Hayman, Balthasar Indermuehle, Kanapathippillai Jeganathan, Simon Johnston, Henry Kanoniuk, Michael Kesteven, Michael Kramer, Mark Leach, Vince Mcintyre, Vanessa Moss, Stefan Osłowski, Chris Phillips, Nathan Pope, Brett Preisig, Daniel Price, Ken Reeves, Les Reilly, John Reynolds, Tim Robishaw, Peter Roush, Tim Ruckley, Elaine Sadler, John Sarkissian, Sean Severs, Ryan Shannon, Ken Smart, Malcolm Smith, Stephanie Smith, Charlotte Sobey, Lister Staveley-Smith, Anastasios Tzioumis, Willem van Straten, Nina Wang, Linqing Wen, Matthew Whiting
-
- Journal:
- Publications of the Astronomical Society of Australia / Volume 37 / 2020
- Published online by Cambridge University Press:
- 08 April 2020, e012
-
- Article
-
- You have access Access
- HTML
- Export citation
-
We describe an ultra-wide-bandwidth, low-frequency receiver recently installed on the Parkes radio telescope. The receiver system provides continuous frequency coverage from 704 to 4032 MHz. For much of the band (
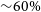 ${\sim}60\%$
), the system temperature is approximately 22 K and the receiver system remains in a linear regime even in the presence of strong mobile phone transmissions. We discuss the scientific and technical aspects of the new receiver, including its astronomical objectives, as well as the feed, receiver, digitiser, and signal processor design. We describe the pipeline routines that form the archive-ready data products and how those data files can be accessed from the archives. The system performance is quantified, including the system noise and linearity, beam shape, antenna efficiency, polarisation calibration, and timing stability.
${\sim}60\%$
), the system temperature is approximately 22 K and the receiver system remains in a linear regime even in the presence of strong mobile phone transmissions. We discuss the scientific and technical aspects of the new receiver, including its astronomical objectives, as well as the feed, receiver, digitiser, and signal processor design. We describe the pipeline routines that form the archive-ready data products and how those data files can be accessed from the archives. The system performance is quantified, including the system noise and linearity, beam shape, antenna efficiency, polarisation calibration, and timing stability.
Contributors
-
- By Maureen Callanan, Eva Chian-Hui Chen, Judy Dunn, Robyn Fivush, Heidi Fung, Joan E. Grusec, Lacey J. Hilliard, Claire Hughes, Deborah Laible, Jin Li, Lynn S. Liben, Megan Luce, Kelly Marin, Natalie Merrill, Peggy J. Miller, Tia Neha, Larry Nucci, Tia Panfile Murphy, Monisha Pasupathi, Holly E. Recchia, Elaine Reese, Jennifer Rigney, Hildy Ross, Qingfang Song, Laura Sterponi, Mele Taumoepeau, Ross A. Thompson, Araceli Valle, Cecilia Wainryb, Qi Wang, Abby C. Winer
- Edited by Cecilia Wainryb, University of Utah, Holly E. Recchia, Concordia University, Montréal
-
- Book:
- Talking about Right and Wrong
- Published online:
- 05 March 2014
- Print publication:
- 20 March 2014, pp xii-xiii
-
- Chapter
- Export citation
Contributors
-
- By Rose Teteki Abbey, K. C. Abraham, David Tuesday Adamo, LeRoy H. Aden, Efrain Agosto, Victor Aguilan, Gillian T. W. Ahlgren, Charanjit Kaur AjitSingh, Dorothy B E A Akoto, Giuseppe Alberigo, Daniel E. Albrecht, Ruth Albrecht, Daniel O. Aleshire, Urs Altermatt, Anand Amaladass, Michael Amaladoss, James N. Amanze, Lesley G. Anderson, Thomas C. Anderson, Victor Anderson, Hope S. Antone, María Pilar Aquino, Paula Arai, Victorio Araya Guillén, S. Wesley Ariarajah, Ellen T. Armour, Brett Gregory Armstrong, Atsuhiro Asano, Naim Stifan Ateek, Mahmoud Ayoub, John Alembillah Azumah, Mercedes L. García Bachmann, Irena Backus, J. Wayne Baker, Mieke Bal, Lewis V. Baldwin, William Barbieri, António Barbosa da Silva, David Basinger, Bolaji Olukemi Bateye, Oswald Bayer, Daniel H. Bays, Rosalie Beck, Nancy Elizabeth Bedford, Guy-Thomas Bedouelle, Chorbishop Seely Beggiani, Wolfgang Behringer, Christopher M. Bellitto, Byard Bennett, Harold V. Bennett, Teresa Berger, Miguel A. Bernad, Henley Bernard, Alan E. Bernstein, Jon L. Berquist, Johannes Beutler, Ana María Bidegain, Matthew P. Binkewicz, Jennifer Bird, Joseph Blenkinsopp, Dmytro Bondarenko, Paulo Bonfatti, Riet en Pim Bons-Storm, Jessica A. Boon, Marcus J. Borg, Mark Bosco, Peter C. Bouteneff, François Bovon, William D. Bowman, Paul S. Boyer, David Brakke, Richard E. Brantley, Marcus Braybrooke, Ian Breward, Ênio José da Costa Brito, Jewel Spears Brooker, Johannes Brosseder, Nicholas Canfield Read Brown, Robert F. Brown, Pamela K. Brubaker, Walter Brueggemann, Bishop Colin O. Buchanan, Stanley M. Burgess, Amy Nelson Burnett, J. Patout Burns, David B. Burrell, David Buttrick, James P. Byrd, Lavinia Byrne, Gerado Caetano, Marcos Caldas, Alkiviadis Calivas, William J. Callahan, Salvatore Calomino, Euan K. Cameron, William S. Campbell, Marcelo Ayres Camurça, Daniel F. Caner, Paul E. Capetz, Carlos F. Cardoza-Orlandi, Patrick W. Carey, Barbara Carvill, Hal Cauthron, Subhadra Mitra Channa, Mark D. Chapman, James H. Charlesworth, Kenneth R. Chase, Chen Zemin, Luciano Chianeque, Philip Chia Phin Yin, Francisca H. Chimhanda, Daniel Chiquete, John T. Chirban, Soobin Choi, Robert Choquette, Mita Choudhury, Gerald Christianson, John Chryssavgis, Sejong Chun, Esther Chung-Kim, Charles M. A. Clark, Elizabeth A. Clark, Sathianathan Clarke, Fred Cloud, John B. Cobb, W. Owen Cole, John A Coleman, John J. Collins, Sylvia Collins-Mayo, Paul K. Conkin, Beth A. Conklin, Sean Connolly, Demetrios J. Constantelos, Michael A. Conway, Paula M. Cooey, Austin Cooper, Michael L. Cooper-White, Pamela Cooper-White, L. William Countryman, Sérgio Coutinho, Pamela Couture, Shannon Craigo-Snell, James L. Crenshaw, David Crowner, Humberto Horacio Cucchetti, Lawrence S. Cunningham, Elizabeth Mason Currier, Emmanuel Cutrone, Mary L. Daniel, David D. Daniels, Robert Darden, Rolf Darge, Isaiah Dau, Jeffry C. Davis, Jane Dawson, Valentin Dedji, John W. de Gruchy, Paul DeHart, Wendy J. Deichmann Edwards, Miguel A. De La Torre, George E. Demacopoulos, Thomas de Mayo, Leah DeVun, Beatriz de Vasconcellos Dias, Dennis C. Dickerson, John M. Dillon, Luis Miguel Donatello, Igor Dorfmann-Lazarev, Susanna Drake, Jonathan A. Draper, N. Dreher Martin, Otto Dreydoppel, Angelyn Dries, A. J. Droge, Francis X. D'Sa, Marilyn Dunn, Nicole Wilkinson Duran, Rifaat Ebied, Mark J. Edwards, William H. Edwards, Leonard H. Ehrlich, Nancy L. Eiesland, Martin Elbel, J. Harold Ellens, Stephen Ellingson, Marvin M. Ellison, Robert Ellsberg, Jean Bethke Elshtain, Eldon Jay Epp, Peter C. Erb, Tassilo Erhardt, Maria Erling, Noel Leo Erskine, Gillian R. Evans, Virginia Fabella, Michael A. Fahey, Edward Farley, Margaret A. Farley, Wendy Farley, Robert Fastiggi, Seena Fazel, Duncan S. Ferguson, Helwar Figueroa, Paul Corby Finney, Kyriaki Karidoyanes FitzGerald, Thomas E. FitzGerald, John R. Fitzmier, Marie Therese Flanagan, Sabina Flanagan, Claude Flipo, Ronald B. Flowers, Carole Fontaine, David Ford, Mary Ford, Stephanie A. Ford, Jim Forest, William Franke, Robert M. Franklin, Ruth Franzén, Edward H. Friedman, Samuel Frouisou, Lorelei F. Fuchs, Jojo M. Fung, Inger Furseth, Richard R. Gaillardetz, Brandon Gallaher, China Galland, Mark Galli, Ismael García, Tharscisse Gatwa, Jean-Marie Gaudeul, Luis María Gavilanes del Castillo, Pavel L. Gavrilyuk, Volney P. Gay, Metropolitan Athanasios Geevargis, Kondothra M. George, Mary Gerhart, Simon Gikandi, Maurice Gilbert, Michael J. Gillgannon, Verónica Giménez Beliveau, Terryl Givens, Beth Glazier-McDonald, Philip Gleason, Menghun Goh, Brian Golding, Bishop Hilario M. Gomez, Michelle A. Gonzalez, Donald K. Gorrell, Roy Gottfried, Tamara Grdzelidze, Joel B. Green, Niels Henrik Gregersen, Cristina Grenholm, Herbert Griffiths, Eric W. Gritsch, Erich S. Gruen, Christoffer H. Grundmann, Paul H. Gundani, Jon P. Gunnemann, Petre Guran, Vidar L. Haanes, Jeremiah M. Hackett, Getatchew Haile, Douglas John Hall, Nicholas Hammond, Daphne Hampson, Jehu J. Hanciles, Barry Hankins, Jennifer Haraguchi, Stanley S. Harakas, Anthony John Harding, Conrad L. Harkins, J. William Harmless, Marjory Harper, Amir Harrak, Joel F. Harrington, Mark W. Harris, Susan Ashbrook Harvey, Van A. Harvey, R. Chris Hassel, Jione Havea, Daniel Hawk, Diana L. Hayes, Leslie Hayes, Priscilla Hayner, S. Mark Heim, Simo Heininen, Richard P. Heitzenrater, Eila Helander, David Hempton, Scott H. Hendrix, Jan-Olav Henriksen, Gina Hens-Piazza, Carter Heyward, Nicholas J. Higham, David Hilliard, Norman A. Hjelm, Peter C. Hodgson, Arthur Holder, M. Jan Holton, Dwight N. Hopkins, Ronnie Po-chia Hsia, Po-Ho Huang, James Hudnut-Beumler, Jennifer S. Hughes, Leonard M. Hummel, Mary E. Hunt, Laennec Hurbon, Mark Hutchinson, Susan E. Hylen, Mary Beth Ingham, H. Larry Ingle, Dale T. Irvin, Jon Isaak, Paul John Isaak, Ada María Isasi-Díaz, Hans Raun Iversen, Margaret C. Jacob, Arthur James, Maria Jansdotter-Samuelsson, David Jasper, Werner G. Jeanrond, Renée Jeffery, David Lyle Jeffrey, Theodore W. Jennings, David H. Jensen, Robin Margaret Jensen, David Jobling, Dale A. Johnson, Elizabeth A. Johnson, Maxwell E. Johnson, Sarah Johnson, Mark D. Johnston, F. Stanley Jones, James William Jones, John R. Jones, Alissa Jones Nelson, Inge Jonsson, Jan Joosten, Elizabeth Judd, Mulambya Peggy Kabonde, Robert Kaggwa, Sylvester Kahakwa, Isaac Kalimi, Ogbu U. Kalu, Eunice Kamaara, Wayne C. Kannaday, Musimbi Kanyoro, Veli-Matti Kärkkäinen, Frank Kaufmann, Léon Nguapitshi Kayongo, Richard Kearney, Alice A. Keefe, Ralph Keen, Catherine Keller, Anthony J. Kelly, Karen Kennelly, Kathi Lynn Kern, Fergus Kerr, Edward Kessler, George Kilcourse, Heup Young Kim, Kim Sung-Hae, Kim Yong-Bock, Kim Yung Suk, Richard King, Thomas M. King, Robert M. Kingdon, Ross Kinsler, Hans G. Kippenberg, Cheryl A. Kirk-Duggan, Clifton Kirkpatrick, Leonid Kishkovsky, Nadieszda Kizenko, Jeffrey Klaiber, Hans-Josef Klauck, Sidney Knight, Samuel Kobia, Robert Kolb, Karla Ann Koll, Heikki Kotila, Donald Kraybill, Philip D. W. Krey, Yves Krumenacker, Jeffrey Kah-Jin Kuan, Simanga R. Kumalo, Peter Kuzmic, Simon Shui-Man Kwan, Kwok Pui-lan, André LaCocque, Stephen E. Lahey, John Tsz Pang Lai, Emiel Lamberts, Armando Lampe, Craig Lampe, Beverly J. Lanzetta, Eve LaPlante, Lizette Larson-Miller, Ariel Bybee Laughton, Leonard Lawlor, Bentley Layton, Robin A. Leaver, Karen Lebacqz, Archie Chi Chung Lee, Marilyn J. Legge, Hervé LeGrand, D. L. LeMahieu, Raymond Lemieux, Bill J. Leonard, Ellen M. Leonard, Outi Leppä, Jean Lesaulnier, Nantawan Boonprasat Lewis, Henrietta Leyser, Alexei Lidov, Bernard Lightman, Paul Chang-Ha Lim, Carter Lindberg, Mark R. Lindsay, James R. Linville, James C. Livingston, Ann Loades, David Loades, Jean-Claude Loba-Mkole, Lo Lung Kwong, Wati Longchar, Eleazar López, David W. Lotz, Andrew Louth, Robin W. Lovin, William Luis, Frank D. Macchia, Diarmaid N. J. MacCulloch, Kirk R. MacGregor, Marjory A. MacLean, Donald MacLeod, Tomas S. Maddela, Inge Mager, Laurenti Magesa, David G. Maillu, Fortunato Mallimaci, Philip Mamalakis, Kä Mana, Ukachukwu Chris Manus, Herbert Robinson Marbury, Reuel Norman Marigza, Jacqueline Mariña, Antti Marjanen, Luiz C. L. Marques, Madipoane Masenya (ngwan'a Mphahlele), Caleb J. D. Maskell, Steve Mason, Thomas Massaro, Fernando Matamoros Ponce, András Máté-Tóth, Odair Pedroso Mateus, Dinis Matsolo, Fumitaka Matsuoka, John D'Arcy May, Yelena Mazour-Matusevich, Theodore Mbazumutima, John S. McClure, Christian McConnell, Lee Martin McDonald, Gary B. McGee, Thomas McGowan, Alister E. McGrath, Richard J. McGregor, John A. McGuckin, Maud Burnett McInerney, Elsie Anne McKee, Mary B. McKinley, James F. McMillan, Ernan McMullin, Kathleen E. McVey, M. Douglas Meeks, Monica Jyotsna Melanchthon, Ilie Melniciuc-Puica, Everett Mendoza, Raymond A. Mentzer, William W. Menzies, Ina Merdjanova, Franziska Metzger, Constant J. Mews, Marvin Meyer, Carol Meyers, Vasile Mihoc, Gunner Bjerg Mikkelsen, Maria Inêz de Castro Millen, Clyde Lee Miller, Bonnie J. Miller-McLemore, Alexander Mirkovic, Paul Misner, Nozomu Miyahira, R. W. L. Moberly, Gerald Moede, Aloo Osotsi Mojola, Sunanda Mongia, Rebeca Montemayor, James Moore, Roger E. Moore, Craig E. Morrison O.Carm, Jeffry H. Morrison, Keith Morrison, Wilson J. Moses, Tefetso Henry Mothibe, Mokgethi Motlhabi, Fulata Moyo, Henry Mugabe, Jesse Ndwiga Kanyua Mugambi, Peggy Mulambya-Kabonde, Robert Bruce Mullin, Pamela Mullins Reaves, Saskia Murk Jansen, Heleen L. Murre-Van den Berg, Augustine Musopole, Isaac M. T. Mwase, Philomena Mwaura, Cecilia Nahnfeldt, Anne Nasimiyu Wasike, Carmiña Navia Velasco, Thulani Ndlazi, Alexander Negrov, James B. Nelson, David G. Newcombe, Carol Newsom, Helen J. Nicholson, George W. E. Nickelsburg, Tatyana Nikolskaya, Damayanthi M. A. Niles, Bertil Nilsson, Nyambura Njoroge, Fidelis Nkomazana, Mary Beth Norton, Christian Nottmeier, Sonene Nyawo, Anthère Nzabatsinda, Edward T. Oakes, Gerald O'Collins, Daniel O'Connell, David W. Odell-Scott, Mercy Amba Oduyoye, Kathleen O'Grady, Oyeronke Olajubu, Thomas O'Loughlin, Dennis T. Olson, J. Steven O'Malley, Cephas N. Omenyo, Muriel Orevillo-Montenegro, César Augusto Ornellas Ramos, Agbonkhianmeghe E. Orobator, Kenan B. Osborne, Carolyn Osiek, Javier Otaola Montagne, Douglas F. Ottati, Anna May Say Pa, Irina Paert, Jerry G. Pankhurst, Aristotle Papanikolaou, Samuele F. Pardini, Stefano Parenti, Peter Paris, Sung Bae Park, Cristián G. Parker, Raquel Pastor, Joseph Pathrapankal, Daniel Patte, W. Brown Patterson, Clive Pearson, Keith F. Pecklers, Nancy Cardoso Pereira, David Horace Perkins, Pheme Perkins, Edward N. Peters, Rebecca Todd Peters, Bishop Yeznik Petrossian, Raymond Pfister, Peter C. Phan, Isabel Apawo Phiri, William S. F. Pickering, Derrick G. Pitard, William Elvis Plata, Zlatko Plese, John Plummer, James Newton Poling, Ronald Popivchak, Andrew Porter, Ute Possekel, James M. Powell, Enos Das Pradhan, Devadasan Premnath, Jaime Adrían Prieto Valladares, Anne Primavesi, Randall Prior, María Alicia Puente Lutteroth, Eduardo Guzmão Quadros, Albert Rabil, Laurent William Ramambason, Apolonio M. Ranche, Vololona Randriamanantena Andriamitandrina, Lawrence R. Rast, Paul L. Redditt, Adele Reinhartz, Rolf Rendtorff, Pål Repstad, James N. Rhodes, John K. Riches, Joerg Rieger, Sharon H. Ringe, Sandra Rios, Tyler Roberts, David M. Robinson, James M. Robinson, Joanne Maguire Robinson, Richard A. H. Robinson, Roy R. Robson, Jack B. Rogers, Maria Roginska, Sidney Rooy, Rev. Garnett Roper, Maria José Fontelas Rosado-Nunes, Andrew C. Ross, Stefan Rossbach, François Rossier, John D. Roth, John K. Roth, Phillip Rothwell, Richard E. Rubenstein, Rosemary Radford Ruether, Markku Ruotsila, John E. Rybolt, Risto Saarinen, John Saillant, Juan Sanchez, Wagner Lopes Sanchez, Hugo N. Santos, Gerhard Sauter, Gloria L. Schaab, Sandra M. Schneiders, Quentin J. Schultze, Fernando F. Segovia, Turid Karlsen Seim, Carsten Selch Jensen, Alan P. F. Sell, Frank C. Senn, Kent Davis Sensenig, Damían Setton, Bal Krishna Sharma, Carolyn J. Sharp, Thomas Sheehan, N. Gerald Shenk, Christian Sheppard, Charles Sherlock, Tabona Shoko, Walter B. Shurden, Marguerite Shuster, B. Mark Sietsema, Batara Sihombing, Neil Silberman, Clodomiro Siller, Samuel Silva-Gotay, Heikki Silvet, John K. Simmons, Hagith Sivan, James C. Skedros, Abraham Smith, Ashley A. Smith, Ted A. Smith, Daud Soesilo, Pia Søltoft, Choan-Seng (C. S.) Song, Kathryn Spink, Bryan Spinks, Eric O. Springsted, Nicolas Standaert, Brian Stanley, Glen H. Stassen, Karel Steenbrink, Stephen J. Stein, Andrea Sterk, Gregory E. Sterling, Columba Stewart, Jacques Stewart, Robert B. Stewart, Cynthia Stokes Brown, Ken Stone, Anne Stott, Elizabeth Stuart, Monya Stubbs, Marjorie Hewitt Suchocki, David Kwang-sun Suh, Scott W. Sunquist, Keith Suter, Douglas Sweeney, Charles H. Talbert, Shawqi N. Talia, Elsa Tamez, Joseph B. Tamney, Jonathan Y. Tan, Yak-Hwee Tan, Kathryn Tanner, Feiya Tao, Elizabeth S. Tapia, Aquiline Tarimo, Claire Taylor, Mark Lewis Taylor, Bishop Abba Samuel Wolde Tekestebirhan, Eugene TeSelle, M. Thomas Thangaraj, David R. Thomas, Andrew Thornley, Scott Thumma, Marcelo Timotheo da Costa, George E. “Tink” Tinker, Ola Tjørhom, Karen Jo Torjesen, Iain R. Torrance, Fernando Torres-Londoño, Archbishop Demetrios [Trakatellis], Marit Trelstad, Christine Trevett, Phyllis Trible, Johannes Tromp, Paul Turner, Robert G. Tuttle, Archbishop Desmond Tutu, Peter Tyler, Anders Tyrberg, Justin Ukpong, Javier Ulloa, Camillus Umoh, Kristi Upson-Saia, Martina Urban, Monica Uribe, Elochukwu Eugene Uzukwu, Richard Vaggione, Gabriel Vahanian, Paul Valliere, T. J. Van Bavel, Steven Vanderputten, Peter Van der Veer, Huub Van de Sandt, Louis Van Tongeren, Luke A. Veronis, Noel Villalba, Ramón Vinke, Tim Vivian, David Voas, Elena Volkova, Katharina von Kellenbach, Elina Vuola, Timothy Wadkins, Elaine M. Wainwright, Randi Jones Walker, Dewey D. Wallace, Jerry Walls, Michael J. Walsh, Philip Walters, Janet Walton, Jonathan L. Walton, Wang Xiaochao, Patricia A. Ward, David Harrington Watt, Herold D. Weiss, Laurence L. Welborn, Sharon D. Welch, Timothy Wengert, Traci C. West, Merold Westphal, David Wetherell, Barbara Wheeler, Carolinne White, Jean-Paul Wiest, Frans Wijsen, Terry L. Wilder, Felix Wilfred, Rebecca Wilkin, Daniel H. Williams, D. Newell Williams, Michael A. Williams, Vincent L. Wimbush, Gabriele Winkler, Anders Winroth, Lauri Emílio Wirth, James A. Wiseman, Ebba Witt-Brattström, Teofil Wojciechowski, John Wolffe, Kenman L. Wong, Wong Wai Ching, Linda Woodhead, Wendy M. Wright, Rose Wu, Keith E. Yandell, Gale A. Yee, Viktor Yelensky, Yeo Khiok-Khng, Gustav K. K. Yeung, Angela Yiu, Amos Yong, Yong Ting Jin, You Bin, Youhanna Nessim Youssef, Eliana Yunes, Robert Michael Zaller, Valarie H. Ziegler, Barbara Brown Zikmund, Joyce Ann Zimmerman, Aurora Zlotnik, Zhuo Xinping
- Edited by Daniel Patte, Vanderbilt University, Tennessee
-
- Book:
- The Cambridge Dictionary of Christianity
- Published online:
- 05 August 2012
- Print publication:
- 20 September 2010, pp xi-xliv
-
- Chapter
- Export citation
Mapping the availability and accessibility of healthy food in rural and urban New Zealand – Te Wai o Rona: Diabetes Prevention Strategy
- Jing Wang, Margaret Williams, Elaine Rush, Nic Crook, Nita G Forouhi, David Simmons
-
- Journal:
- Public Health Nutrition / Volume 13 / Issue 7 / July 2010
- Published online by Cambridge University Press:
- 28 September 2009, pp. 1049-1055
-
- Article
-
- You have access Access
- HTML
- Export citation
-
Objective
Uptake of advice for lifestyle change for obesity and diabetes prevention requires access to affordable ‘healthy’ foods (high in fibre/low in sugar and fat). The present study aimed to examine the availability and accessibility of ‘healthy’ foods in rural and urban New Zealand.
DesignWe identified and visited (‘mapped’) 1230 food outlets (473 urban, 757 rural) across the Waikato/Lakes areas (162 census areas within twelve regions) in New Zealand, where the Te Wai O Rona: Diabetes Prevention Strategy was underway. At each site, we assessed the availability of ‘healthy’ foods (e.g. wholemeal bread) and compared their cost with those of comparable ‘regular’ foods (e.g. white bread).
ResultsHealthy foods were generally more available in urban than rural areas. In both urban and rural areas, ‘healthy’ foods were more expensive than ‘regular’ foods after adjusting for the population and income level of each area. For instance, there was an increasing price difference across bread, meat, poultry, with the highest difference for sugar substitutes. The weekly family cost of a ‘healthy’ food basket (without sugar) was 29·1 % more expensive than the ‘regular’ basket ($NZ 176·72 v. $NZ 136·84). The difference between the ‘healthy’ and ‘regular’ basket was greater in urban ($NZ 49·18) than rural areas ($NZ 36·27) in adjusted analysis.
Conclusions‘Healthy’ foods were more expensive than ‘regular’ choices in both urban and rural areas. Although urban areas had higher availability of ‘healthy’ foods, the cost of changing to a healthy diet in urban areas was also greater. Improvement in the food environment is needed to support people in adopting healthy food choices.
DIVISION VIII / WORKING GROUP SUPERNOVA
- Wolfgang Hillebrandt, Brian P. Schmidt, Edward Baron, Stefano Benetti, Sergey I. Blinnikov, Sergey I. Blinnikov, David R. Branch, Enrico Cappellaro, Alexei V. Filippenko, Claes Fransson, Peter M. Garnavich, Daniel W. E. Green, Ariel M. Goobar, Mario Hamuy, Peter H. Hauschildt, Robert P. Kirshner, Bruno Leibundgut, Daniel J. Lennon, Eric J. Lentz, Peter Lundqvist, Robert McGraw, Paolo A. Mazzali, W. Peter S. Meikle, Anthony Mezzacappa, Jens C. Niemeyer, Ken'ichi Nomoto, Reynald Pain, Nino Panagia, Ferdinando Patat, Mark M. Phillips, Elena Pian, Guiliano Pignata, Philipp Podsiadlowski, María Pilar Ruiz-Lapuente, Elaine M. Sadler, Brian P. Schmidt, Peter O. Shull, Jason Spyromilio, Nicholas B. Suntzeff, Friedrich-Karl Thielemann, Christopher Tout, Virginia L. Trimble, James W. Truran, Dmitry Yu. Tsvetkov, Massimo Turatto, Massimo della Valle, Schuyler Van Dyk, Wolfgang H. Voges, Nicholas A. Walton, Lifan Wang, J. Craig Wheeler, Kurt W. Weiler, Patricia A. Whitelock, Stanford E. Woosley, Hitoshi Yamaoka, Gang Zhao
-
- Journal:
- Proceedings of the International Astronomical Union / Volume 4 / Issue T27A / December 2008
- Published online by Cambridge University Press:
- 01 December 2008, pp. 295-297
- Print publication:
- December 2008
-
- Article
-
- You have access Access
- Export citation
-
The Supernova Working Group was re-established at the IAU XXV General Assembly in Sydney, 21 July 2003, sponsored by Commissions 28 (Galaxies) and 47 (Cosmology). Here we report on some of its activities since 2005.
18 - Inflammation and stroke: benefits or harm?
- from Part VI - Inflammation
-
- By Giora Z. Feuerstein, DuPont Pharmaceuticals Company, Wilmington, DE, Elaine E. Peters, DuPont Pharmaceuticals Company, Wilmington, DE, Xinkang Wang, DuPont Pharmaceuticals Company, Wilmington, DE
- Edited by Pak H. Chan, Stanford University, California
-
- Book:
- Cerebrovascular Disease
- Published online:
- 02 November 2009
- Print publication:
- 28 March 2002, pp 213-225
-
- Chapter
- Export citation
-
Summary
Introduction
Stroke is the third leading cause of death in most developed countries and the primary cardiovascular cause of death in Japan and China. The health burden of the disease is staggering, as loss of a productive life inflicts a heavy toll on patients, families and the society at large. Yet, this disease has no effective therapeutic treatment beyond a limited (about 2%) treatment with thrombolytics that has a significant adverse effect, and despite intense research efforts and numerous clinical trials to develop drugs to reduce morbidity and mortality. Over the past two decades, the focus of drug development efforts has targeted modulators of ion channels (Ca2+, Na+), scavengers of oxygen radicals and antagonists of excitotoxic neurotransmitters (primarily glutamate and glycine receptors). However, all phase III clinical trials with these compounds (Table 18.1), where such compounds were tested for efficacy of treatment in ischemic stroke, have failed due to lack of efficacy, adverse effects or other development difficulties. Debates on the reasons for this grim reality have sprung up in recent meetings, where “finger pointing” to possible causes of failure include wrong animal models, wrong mechanism of action, poor clinical design, inadequate timing of treatment, etc. While this debate is ongoing, the stroke research community seems to have been disenchanted by the “classic” targets for drug development (vide supra) as evidenced by emerging hope that other strategies such as “reconstruction”, “apoptosis” and “inflammation” may yield the desired success.
Staphylococcus aureus Colonization and Infection After Discharge from a Term Newborn Nursery
- Elaine E.L. Wang, Diane Elder, Norma Mishkel
-
- Journal:
- Infection Control / Volume 8 / Issue 1 / January 1987
- Published online by Cambridge University Press:
- 02 January 2015, pp. 30-33
- Print publication:
- January 1987
-
- Article
- Export citation
-
During a 6-month period, term infants underwent nasal and umbilical swabbing within 96 hours of delivery and again at 1 to 18 (mean 3.8) weeks after discharge. Swabs were inoculated onto horse blood agar and all S aureus isolates were phage typed. Two hundred three infants were enrolled and follow-up was obtained for 181 (89%). Thirty-two of 181 (17.6%) were initially colonized, of whom 12 (37.5%) were colonized on follow-up. Thirty-two of 181 became colonized subsequent to hospital discharge. No single phage type predominated. Twenty-five patients were diagnosed to have clinical illness—omphalitis (11), conjunctivitis (10), and pustulosis (5). Six of these were colonized with 5 aureus initially, although organisms of the same phage type were not recovered from cultures obtained at the time of clinical illness. There was no significant difference in the rate of infections in colonized (19%) versus noncolonized (12%) infants. In five patients where S aureus was recovered at the time of symptoms, all organisms were acquired subsequent to discharge. We conclude that nursery colonization with S aureus did not lead to clinical illness, and clinical illnesse s previously ascribed to S aureus frequently occur in the absence of these bacteria.